The Rocky Journey to Accessible Male Birth Control
If you’re eagerly awaiting more contraceptive options for men, stop holding your breath.
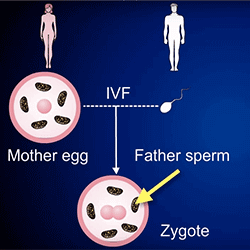
Excitement is growing among men and women alike about the impending availability of male birth control. From pills to topical gels to injections, there are several contraceptives for men in development.
Yet despite hopeful reports from scientists and the media, the reality is it may still be a long time before male birth control becomes available to consumers.
When will male birth control be available?
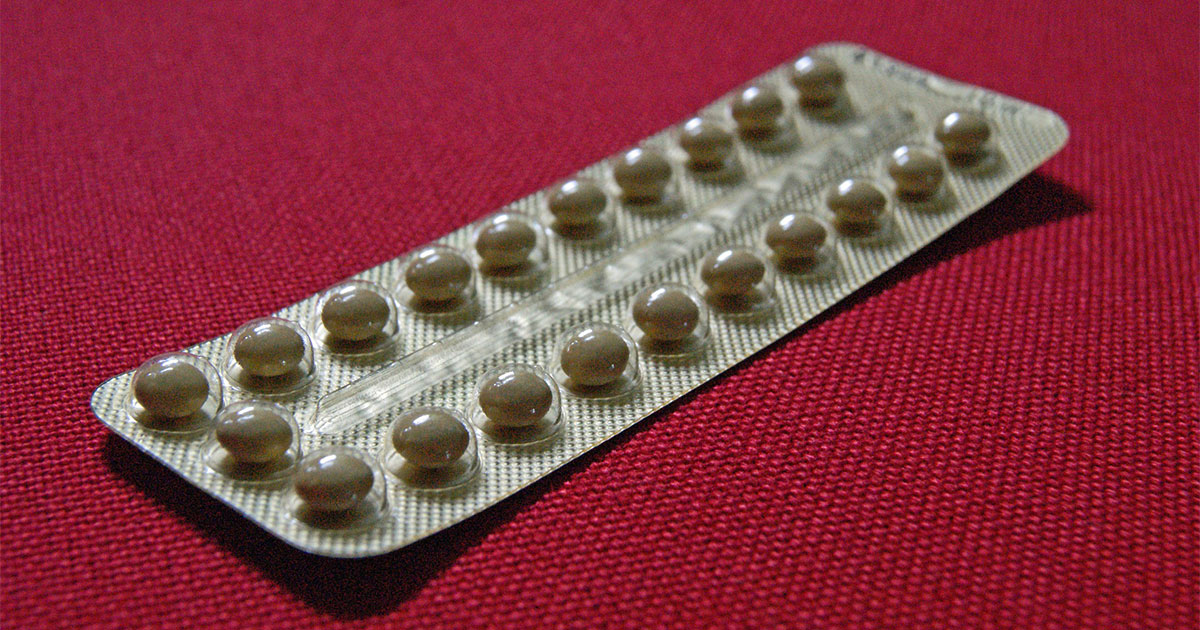
Currently, the only available options for male birth control are condoms and vasectomies. At this time, there is no definitive timeline for when any of the male contraceptive options in development will be available to the public.
The male birth control pill has been in development for more than 60 years and will likely be the first method available to consumers.
But when it will become available remains a mystery.
In March 2017, Airlangga University in Indonesia announced their partnership with Harsen Laboratories and plans to distribute the world’s first male birth control pill by September 2018.
This announcement was preceded by two earlier announcements touting the drug’s impending release in 2013 and 2015 respectively. As of this writing, the drug is still not available and no public updates have been provided since 2017.
The troubled history of the male pill
The male birth control pill has been in development since 1957, when endocrinologist Gregory Pincus, co-creator of the female birth control pill, first tested the same approach on men.

Dr. Elsimar Coutinho
Unfortunately, interest in this side of Pincus’ research was low at the time, given the enormous popularity of the female pill.
In the 1960s, Brazilian fertility doctor Elsimar Coutinho began working with the Chinese government on the development of gossypol, a non-hormonal method of oral contraception for men.
Clinical trials showed gossypol pills successfully reduced male sperm count. However, the results were permanent for many study participants as sperm levels did not return to normal after they stopped taking the drug.
Further studies conducted over the next three decades concluded that over time, gossypol also caused damage to the lining of the ducts that store sperm. In 1998, this research was abandoned after the World Health Organization declared gossypol unsuitable for male contraceptive use due to the toxic side effects and risk of infertility.
Another successful but halted attempt
In the late 1970s, the National Institutes of Health began clinical trials for a new, injectable method of male birth control. These trials resulted in several studies proving that testosterone injections for male contraception were just as effective as female contraception.
Further studies over the next three decades confirmed the efficacy and reversibility of this method. However, the most recent trial, backed by the World Health Organization and the Canadian Oilsands Network for Research and Development (CONRAD), was terminated prematurely by an independent safety review panel due to the frequency and severity of adverse side effects.
The study results published in 2016 listed the following as the most common side effects: acne, injection site pain, increased libido, and mood disorders. Two serious adverse events were reported by men in the study including the attempted suicide of one participant (out of 320 total participants), which ultimately led to the termination of the trial.
To compare, the most common side effects suffered by women using birth control pills include: nausea, headaches, weight gain, decreased libido, mood changes, vaginal dryness, irregular bleeding, blood clots, seizures, heart conditions, abdominal pain, blurred vision, and in rare cases death.
Obstacles to reaching the market
In order for any of the male contraceptive methods in development to become available for purchase at the pharmacy, scientists must first conduct human clinical trials, which can be quite expensive.
“We’re talking about drugs men are going to take for a really long time, so the pathway for approval is long too,” explains Diana Blithe, program director for contraception development at the U.S. National Institute of Child Health and Human Development (NICHD).
Before a drug is eligible for an FDA application, three phases of clinical trials must be conducted. Scientists in the field of male contraception have historically struggled with declining interest from pharmaceutical companies to fund the trials needed to obtain FDA approval.
According to the Male Contraceptive Initiative, only “10% of drugs [in development], on average, make it from Phase I to market.”
Lack of funding and commercial interest
Despite the estimated global market value of up to $200 billion and numerous multicultural studies citing the willingness of men to use male contraception, there is still a diminutive interest from pharmaceutical companies to support male contraception research. Financial support from nonprofit and philanthropic organizations, as well as government agencies continues, but at decreased levels.
In the last fifteen years, three major pharmaceutical companies have pulled the plug on research programs focused on male contraception. Companies have indicated these decisions were motivated by “changes in corporate direction.”
In an interview with Mosaic, Coelingh Bennink, a former executive on one of the terminated studies, provides insight into the executive atmosphere and attitudes that could be driving corporate perceptions:
At board level it was only middle-aged white males… I tried to explain how important [male contraceptive research] could be, but they never got further than saying to each other, ‘Would you do it?’ ‘No, I wouldn’t do it.’ It was not considered male behavior to take responsibility for contraception.
Image sources: Anqa, fotos GOVBA
Leave a reply
You must be logged in to post a comment.